 For example Sodium in period 3 has an atomic radius of 186 x10-12 m and chlorine in the same period has an atomic radius of 99x10-12 m as Chlorine has a larger number of protons and a higher nuclear charge to attract the electrons to the nucleus and make the radius smaller. This means the electrons are pulled in closer to the nucleus reducing the size of the atomic radius. At the same time when elements are in the same period they have the same number of shells, this means the shielding of this layer of electrons is the same for all elements in a period but the number of protons and nuclear charge increases. For example the atomic radius of Lithium is 152x10-12 m but if we progress down to caesium the atomic radius of caesium is 262x10-12m as caesium has 6 electron shells and also a greater number of protons but due to the large number of shells the outer valence electron is much further away.Īcross a period the atomic radius decreases as the number of protons in the nucleus increases, this means the nucleus has a higher nuclear charge and can attract electrons more closely due to the attraction of the positive protons to the negative electrons. These five toxic, non-metallic elements make up Group 17 of the periodic table and consist of: fluorine (F), chlorine (Cl), bromine (Br), iodine (I), and astatine (At). Atomic size effects upon the strength of SRO are not very significant at room temperature but they do appear to determine the extent of the athermal regime, making them crucial for the high temperature strength. In general as we progress down the group the number of electrons and shells increases and so does the size of the nucleus meaning atomic radius will increase as we progress down a group. Atomic and Physical Properties of Halogens Halogen Group (Group 17) Trends The halogens are located on the left of the noble gases on the periodic table. Atomic radius is the distance from the atoms nucleus to the outermost electron orbital and a lot of trends in the periodic table rely on this property due to its relation to nuclear charge and shielding. As the name portends, these are empirical radii obtained from measured bond lengths. from the nucleus to the outermost shell (valence shell) of that atom. We briefly describe five of its accepted measures. We will use the covalent radius ( Figure 4.3.1 ), which is defined as one-half the distance between the nuclei of two identical atoms when they are joined by a covalent bond (this measurement is possible because atoms within molecules still retain much of their atomic identity). Atomic size is the distance between the center of an atom i.e. In this section, we discuss how atomic and ion sizes are defined and obtained. 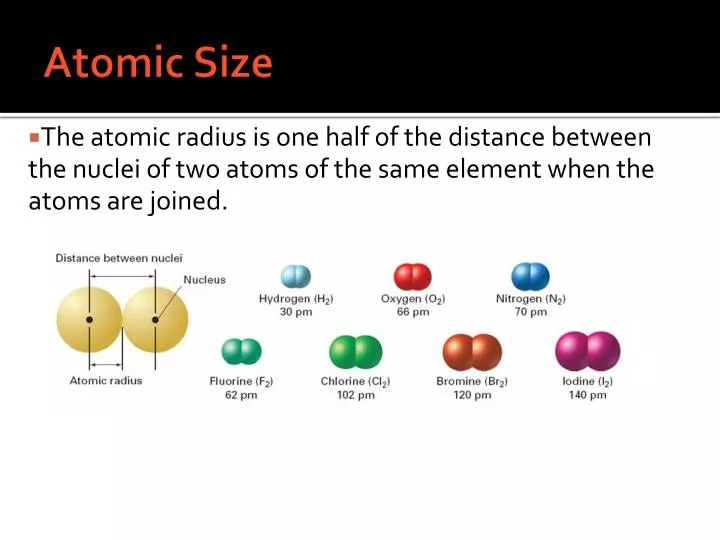 
The atomic radius of an atom is useful to understand as it helps to understand many properties of atoms and how they react. Atomic size is a qualitative concept that is fundamental to the study of chemistry. As a result, atoms and ions cannot be said to have exact sizes however, some atoms are larger or smaller than others, and this influences their chemistry. 
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
